
- #ISO 13485 TRAINING ONLINE HOW TO#
- #ISO 13485 TRAINING ONLINE REGISTRATION#
- #ISO 13485 TRAINING ONLINE SOFTWARE#
- #ISO 13485 TRAINING ONLINE ISO#
- #ISO 13485 TRAINING ONLINE PLUS#
#ISO 13485 TRAINING ONLINE ISO#
The course is made for beginners in quality management and ISO standards, and no prior knowledge is needed to take this course.Īfter passing the exam, you will receive a certificate proving that you have a working knowledge of all ISO 13485 requirements.
#ISO 13485 TRAINING ONLINE SOFTWARE#
Ask us to schedule a demo so you can explore a software solution that connects with your processes, people, and data.In this online course, you’ll learn everything you need to know about ISO 13485, including all the requirements and best practices for compliance. This helps you leverage an eQMS system designed specifically for the medical device industry.
#ISO 13485 TRAINING ONLINE REGISTRATION#
Optional: Additional days may be added to incorporate how the standard relates to other international regulations, including registration requirements in various markets like Canada, Europe, Australia, and others.Įmergo by UL can provide eQMS support in strategic alliance with Greenlight Guru.
#ISO 13485 TRAINING ONLINE HOW TO#
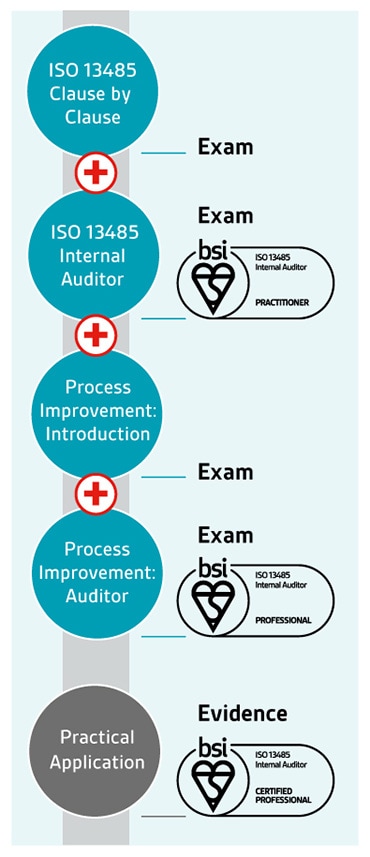

Improve your company's understanding of why the process approach works.Identify key personnel inside your company who will be involved in implementation or transition, and maintenance.The responsibility and commitment of senior management, as well as the participation from company employees.Learn how changes may impact your organization and its current QMS.Benefits of adopting or upgrading to the ISO 13485:2016 standard, and its relationship to EN ISO 13485:2016.

Overview of the ISO 13485:2016 standard and the implications to your company.What your team will learn in this intensive one-day ISO 13485 awareness training class: On-site training allows your employees to ask questions, something they cannot do easily with online classes, and ensures every employee receives consistent information. Our training is a great value for companies training two or more employees at a time.
#ISO 13485 TRAINING ONLINE PLUS#
Our fee for ISO 13485:2016 training is based on a set daily rate plus travel expenses. Customized on-site ISO 13485:2016 training for a fixed price, regardless of how many employees attend. Upgrading to ISO 13485:2016? See how we can help. Your team will receive an in-depth ISO 13485 training session tailored to the needs of your organization. It is conducted on-site by a senior consultant with extensive, real-world auditing and manufacturing experience across various device types and quality systems. The new ISO 13485:2016 focuses on the entire supply chain of the medical device industry, with emphasis on risk management.Įmergo's ISO 13485:2016 employee training class provides your employees with an overview of ISO 13485 and their responsibilities. For your QMS to succeed, it is critical that your employees understand the necessary procedures and daily functions. Implementation and maintenance of your ISO 13485:2016 quality management system requires the participation of your entire team.
